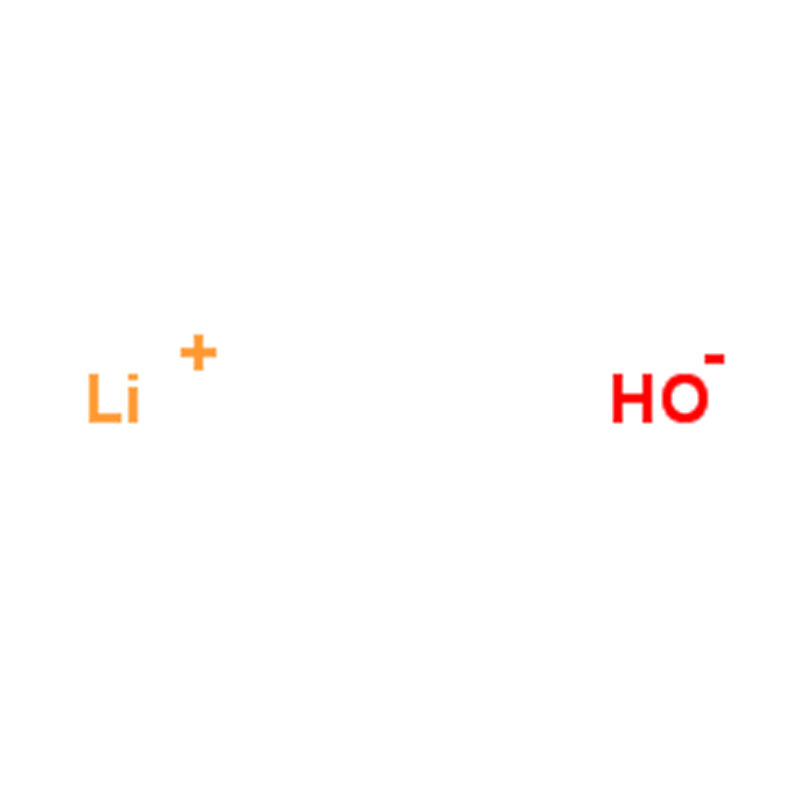
Lithium hydroxide CAS#1310-65-2
High Purity Crystalline Forms
Available as white tetragonal anhydrous crystals and monoclinic monohydrate crystals, ensuring consistent quality and reliability.
Excellent Thermal Stability
Anhydrous lithium hydroxide melts at 450°C and decomposes at 924°C, suitable for high-temperature industrial processes.
Good Solubility in Water
Both forms are water-soluble, with the monohydrate showing higher solubility, allowing flexible use in aqueous applications.
Controlled Solubility in Organic Solvents
Slightly soluble in alcohol and insoluble in ether, enabling predictable chemical behavior in mixed-solvent systems.
Product Description of Lithium Hydroxide CAS#1310-65-2
Lithium hydroxide appears as white tetragonal crystals with a refractive index of 1.464 and a density of 1.46 g/cm³. It melts at 450°C and decomposes at 924°C. The compound is soluble in water (12.8 g/100 g at 20°C, 17.5 g/100 g at 100°C) and slightly soluble in alcohol.
Lithium hydroxide monohydrate is a white monoclinic crystalline solid with a refractive index of 1.460 and a density of 1.51 g/cm³. It is soluble in water, with greater solubility than the anhydrous form (22.3 g/100 g at 10°C, 26.8 g/100 g at 100°C), slightly soluble in alcohol, and insoluble in ether.
Lithium hydroxide Chemical Properties
| Melting point | 470 °C (dec.) (lit.) |
| Boiling point | 925°C |
| density | 1.43 |
| bulk density | 550kg/m3 |
| storage temp. | Store below +30°C. |
| solubility | water: soluble71g/L at 20°C |
| form | Solid |
| pka | 14[at 20 ℃] |
| Specific Gravity | 2.54 |
| color | White to light yellow |
| Odor | odorless |
| PH | 12 (50g/l, H2O, 50℃) |
| Water Solubility | 113 g/L (20 ºC) |
| Sensitive | Air Sensitive & Hygroscopic |
| Merck | 145,534 |
| Stability: | Stable. Incompatible with moisture. strong acids, carbon dioxide. |
| Cosmetics Ingredients Functions | HAIR WAVING OR STRAIGHTENING |
| BUFFERING | |
| InChIKey | WMFOQBRAJBCJND-UHFFFAOYSA-M |
| CAS DataBase Reference | 1310-65-2(CAS DataBase Reference) |
| NIST Chemistry Reference | Lithium hydroxide(1310-65-2) |
| EPA Substance Registry System | Lithium hydroxide (1310-65-2) |
Safety Information
| Hazard Codes | C |
| Risk Statements | 52/53-35-20/22-22-34-23 |
| Safety Statements | 45-36/37/39-26-22 |
| RIDADR | 2680 |
| WGK Germany | 1 |
| RTECS | OJ6307070 |
| TSCA | TSCA listed |
| HazardClass | 8 |
| PackingGroup | II |
| HS Code | 28252010 |
| Toxicity | LD50 orally in Rabbit: 210 mg/kg |
Product Application of Lithium Hydroxide CAS#1310-65-2
Lithium hydroxide is widely used as a heat transfer medium and as an electrolyte in storage batteries. It is also applied in the production of lithium greases.
Additionally, it is used in ceramics, certain Portland cement formulations, and for the absorption of carbon dioxide in sealed environments such as submarines, spacecraft, and breathing apparatus. Lithium hydroxide is further employed in esterification reactions, as a stabilizer in photographic development, and as a coolant in pressurized water reactors to control corrosion.
Factory and Equipment Show