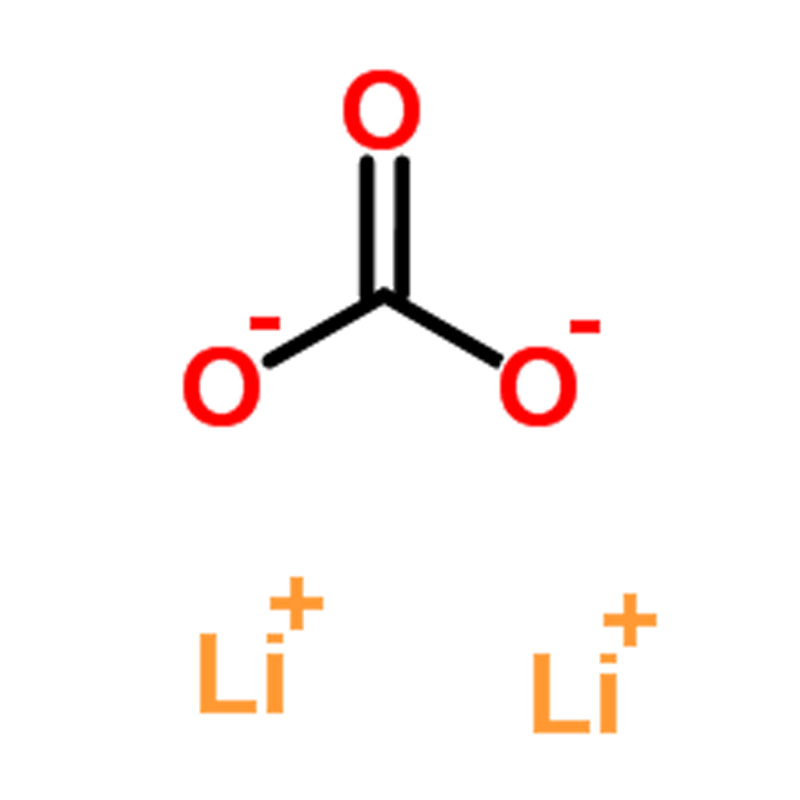
Product Description of Lithium Carbonate CAS#554-13-2
Lithium carbonate (Li₂CO₃) is a colorless monoclinic crystal or white powder. It has a density of 2.11 g/cm³ and a melting point of 618℃. The compound is non-deliquescent and stable in air. It has low solubility in water, which decreases as the temperature increases.