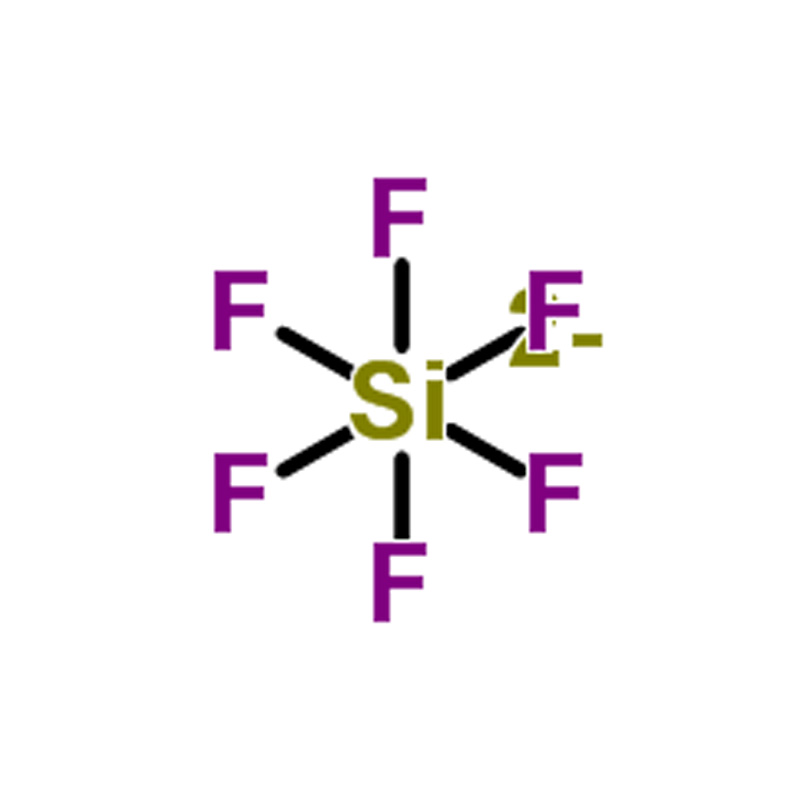
Hexafluorosilicic acid CAS#16961-83-4
Wide Industrial Applicability
Suitable for use across water treatment, chemical manufacturing, metallurgy, and organic synthesis, offering broad application versatility.
Key Raw Material for Fluoride Compounds
An essential precursor for the production of aluminum fluoride, cryolite, and various hexafluorosilicate salts, supporting stable downstream manufacturing.
Functional Performance in Metallurgy
Effectively used as an electrolyte in the Betts electrolytic process, contributing to efficient and reliable lead refining operations.
High Reactivity in Chemical Synthesis
Acts as a specialized reagent for selective Si–O bond cleavage in silyl ethers, enabling precise control in organic synthesis processes.
Product Application of Hexafluorosilicic Acid CAS#16961-83-4
Hexafluorosilicic acid is an inorganic acid widely applied in multiple industrial fields. It is primarily used in water fluoridation programs in the United States to help reduce the occurrence of dental caries and dental fluorosis. In chemical manufacturing, it serves as a key raw material for producing aluminum fluoride, cryolite, and various hexafluorosilicate salts. Additionally, it is utilized in the production of silicon and silicon dioxide. The product also functions as an electrolyte in the Betts electrolytic process for lead refining. In organic synthesis, hexafluorosilicic acid is employed as a specialized reagent for the selective cleavage of Si–O bonds in silyl ethers.
Hexafluorosilicic acid Chemical Properties
| Boiling point | 108-109°C |
| density | 1.22 g/mL at 20 °C (lit.) 1.31 g/mL at 25 °C |
| vapor pressure | 23hPa at 19.85℃ |
| refractive index | 1.35 |
| Fp | 108-109°C |
| storage temp. | −20°C |
| solubility | H2O: 1 mg/mL, clear, colorless |
| form | Liquid |
| pka | 1.83[at 20 ℃] |
| color | Clear colorless |
| Specific Gravity | 1.38 (40%) |
| Water Solubility | Miscible with water. |
| Hydrolytic Sensitivity | 0: forms stable aqueous solutions |
| Merck | 144,182 |
| Exposure limits | ACGIH: TWA 2.5 mg/m3 |
| NIOSH: IDLH 250 mg/m3; TWA 2.5 mg/m3 | |
| Stability: | Stable in aqueous solution. |
| InChI | 1S/F4Si.2FH/c1-5(2,3)4;;/h;2*1H |
| InChIKey | AUJBMDCSBIPDEH-UHFFFAOYSA-N |
| SMILES | F.F.F[Si](F)(F)F |
| CAS DataBase Reference | 16961-83-4(CAS DataBase Reference) |
| EPA Substance Registry System | Fluosilicic acid (16961-83-4) |
Safety Information
| Hazard Codes | C |
| Risk Statements | 34-35-20/21/22 |
| Safety Statements | 26-36/37/39-45-27 |
| RIDADR | UN 1778 8/PG 2 |
| WGK Germany | 3 |
| RTECS | VV8225000 |
| F | 8月10日 |
| Hazard Note | Corrosive |
| TSCA | TSCA listed |
| HazardClass | 8 |
| PackingGroup | II |
| HS Code | 28111990 |
| Storage Class | 6.1D - Non-combustible acute toxic Cat.3 |
| toxic hazardous materials or hazardous materials causing chronic effects | |
| Hazard Classifications | Acute Tox. 3 Dermal |
| Acute Tox. 4 Oral | |
| Eye Dam. 1 | |
| Skin Corr. 1 | |
| Hazardous Substances Data | 16961-83-4(Hazardous Substances Data) |
| Toxicity | LD50 oral in rat: 430mg/kg |
Product Application of Hexafluorosilicic Acid CAS#16961-83-4
Hexafluorosilicic acid is widely utilized as a fluoride source and can be processed into various valuable hexafluorosilicate salts. It also serves as an electrolyte in the Betts electrolytic process for lead refining. In organic chemistry, it functions as an important reagent for the cleavage of Si–O bonds in silyl ethers. In addition, the product is applied as a wood preservative and is used for the surface modification of calcium carbonate.
Factory and Equipment Show