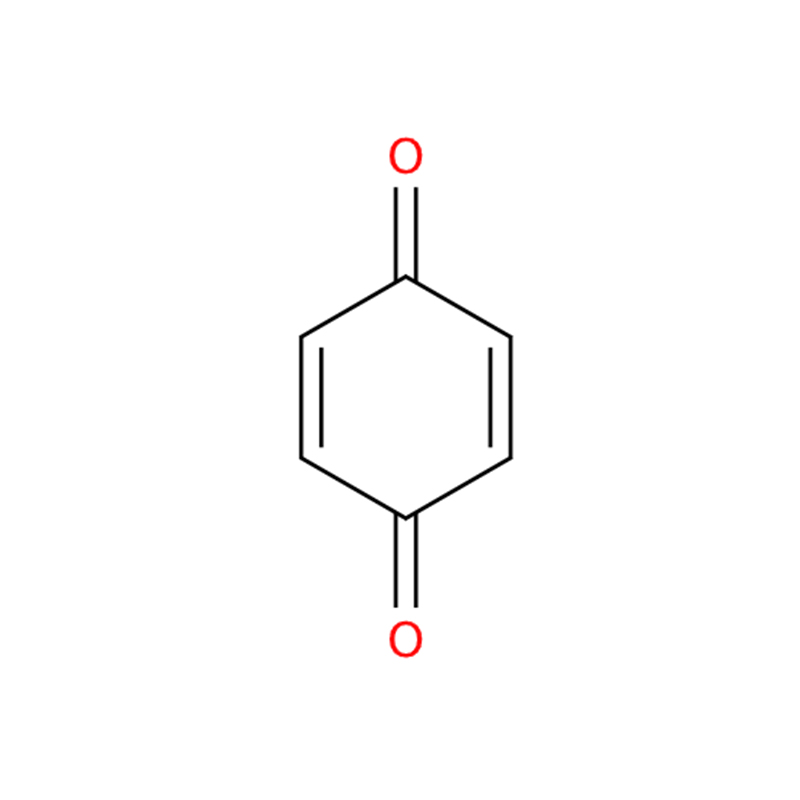
1,4-Benzoquinone CAS#106-51-4
Versatile Chemical Intermediate – Widely used in the production of hydroquinone, rubber accelerators, oxidizing agents, and other chemicals.
Polymerization Inhibitor – Effective in preventing unwanted polymerization in chemical processes.
Broad Industrial Applications – Utilized in the dye, textile, tanning, cosmetic, and photographic industries.
Oxidizing Agent – Serves as a reliable oxidizing agent in various chemical reactions and laboratory applications.
1,4-Benzoquinone CAS#106-51-4
Quinone (p-benzoquinone) appears as a large yellow monoclinic prism with a strong, chlorine-like odor. It is widely used as a chemical intermediate, polymerization inhibitor, oxidizing agent, photographic chemical, tanning agent, and general chemical reagent. First commercially produced in 1919, it has since been manufactured in several European countries. Its primary application is in hydroquinone production, but it also serves as an intermediate for rubber accelerators, oxidizing agents, and other chemical products. Quinone finds applications across the dye, textile, chemical, tanning, and cosmetic industries, as well as in chemical synthesis, manufacturing processes, and laboratories involving protein fibers, photographic films, hydrogen peroxide, and gelatin production. Occupational and inhalation exposure may occur in industrial settings and from sources such as tobacco smoke.
1,4-Benzoquinone Chemical Properties
| Melting point | 113-115 °C(lit.) |
| Boiling point | 293°C |
| bulk density | 700kg/m3 |
| density | 1.31 |
| vapor density | 3.73 (vs air) |
| vapor pressure | 0.1 mm Hg ( 25 °C) |
| refractive index | n20/D 1.453 |
| Fp | 38°C |
| storage temp. | room temp |
| solubility | 10g/l |
| pka | 7.7 |
| form | Powder |
| color | Yellow to green |
| Odor | irritant odor |
| PH | 4 (1g/l, H2O, 20℃) |
| biological source | synthetic |
| Water Solubility | 10 g/L (25 ºC) |
| Merck | 148074 |
| BRN | 773967 |
| Exposure limits | TLV-TWA 0.4 mg/m3 (0.1 ppm); STEL 1.2 mg/m3 (0.3 ppm) (ACGIH); IDLH 75 ppm (NIOSH). |
| Stability: | Stable, but light sensitive. Incompatible with strong oxidizing agents. Flammable. |
| InChI | 1S/C6H4O2/c7-5-1-2-6(8)4-3-5/h1-4H |
| InChIKey | AZQWKYJCGOJGHM-UHFFFAOYSA-N |
| SMILES | O=C1C=CC(=O)C=C1 |
| LogP | 0.1-0.3 at 23℃ and pH4.8-5.3 |
| CAS DataBase Reference | 106-51-4(CAS DataBase Reference) |
| NIST Chemistry Reference | p-Benzoquinone(106-51-4) |
| IARC | 3 (Vol. 15, Sup 7, 71) 1999 |
| EPA Substance Registry System | Quinone (106-51-4) |
Safety Information
| Hazard Codes | T,N,Xn,F |
| Risk Statements | 23/25-36/37/38-50-20/21/22-11 |
| Safety Statements | 26-28-45-61-28A-23-16 |
| RIDADR | UN 2587 6.1/PG 2 |
| OEB | C |
| OEL | TWA: 0.4 mg/m3 (0.1 ppm) |
| WGK Germany | 3 |
| RTECS | DK2625000 |
| F | 8 |
| Autoignition Temperature | 815 °F |
| TSCA | TSCA listed |
| HS Code | 2914 69 80 |
| HazardClass | 6.1 |
| PackingGroup | II |
| Storage Class | 4.1B - Flammable solid hazardous materials |
| Hazard Classifications | Acute Tox. 3 Inhalation |
| Acute Tox. 3 Oral | |
| Aquatic Acute 1 | |
| Aquatic Chronic 1 | |
| Eye Dam. 1 | |
| Flam. Sol. 1 | |
| Muta. 2 | |
| Skin Corr. 1B | |
| Skin Sens. 1 | |
| STOT SE 3 | |
| Hazardous Substances Data | 106-51-4(Hazardous Substances Data) |
| Toxicity | LD50 orally in rats: 130 mg/kg (Woodard) |
| IDLA | 100 mg/m3 |
Product Application of 1,4-Benzoquinone CAS#106-51-4
p-Benzoquinone is employed as a dienophile in Diels-Alder cycloadditions for the synthesis of naphthoquinones and 1,4-phenanthrenediones. It serves as a dehydrogenation reagent and oxidizing agent in synthetic organic chemistry. In the Thiele-Winter reaction, it participates in the preparation of triacetate of hydroxyquinol via reaction with acetic anhydride and sulfuric acid. Additionally, it is used in the synthesis of bromadol and to suppress double-bond migration during olefin metathesis reactions. p-Benzoquinone also acts as a precursor to hydroquinone, which has applications in photography, and functions as a reducing agent and antioxidant in rubber production.