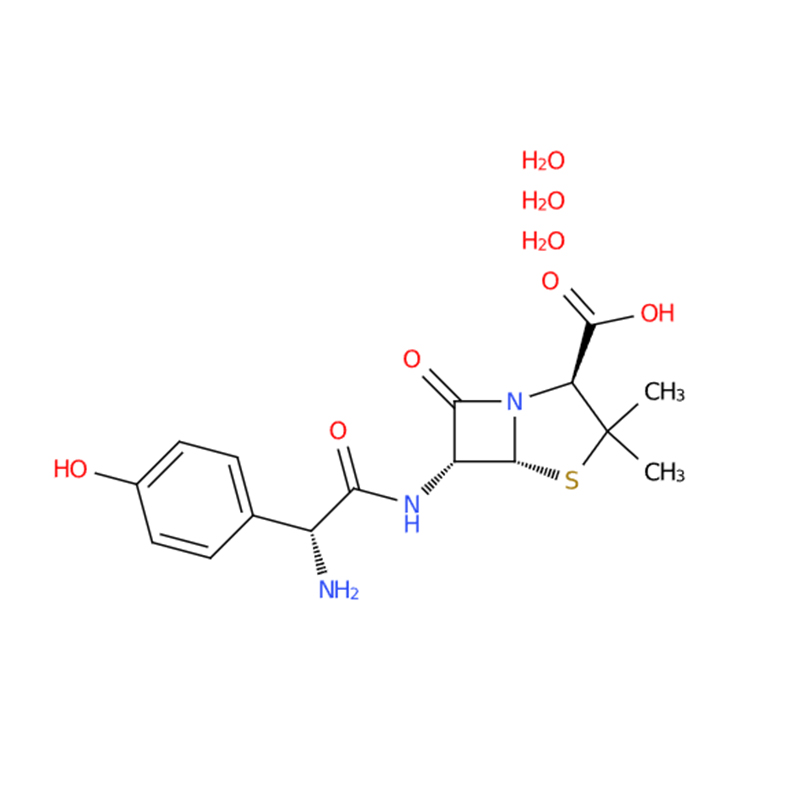
Amoxicillin trihydrate CAS#61336-70-7
Broad-Spectrum Antibacterial Activity: As a semi-synthetic broad-spectrum penicillin, amoxicillin is effective against a wide range of susceptible bacterial infections.
Reliable Therapeutic Efficacy: It delivers proven therapeutic effects consistent with ampicillin, ensuring dependable performance in pharmaceutical formulations.
Versatile Pharmaceutical Applications: The product is widely used in various pharmaceutical preparations for both human and veterinary antibacterial treatments.
Compliant with EU Regulatory Standards: It conforms to Commission Regulation (EU) No 37/2010 regarding pharmacologically active substances and maximum residue limits for foods of animal origin.
Products Description of Amoxicillin trihydrate CAS#61336-70-7
Amoxicillin trihydrate (CAS# 61336-70-7) is a white-to-white-orange crystalline powder, semi-synthetic, broad-spectrum-lactam antibiotic used to treat bacterial infections. It is a trihydrate form with $\geq$98% purity, acting by inhibiting bacterial cell wall synthesis.
Parameters
| Melting point | >200°C (dec.) |
| alpha | D20 +246° (c = 0.1) |
| refractive index | 302 ° (C=0.1, H2O) |
| storage temp. | Inert atmosphere,2-8°C |
| solubility | Slightly soluble in water, very slightly soluble in ethanol (96 per cent), practically insoluble in fatty oils. It dissolves in dilute acids and dilute solutions of alkali hydroxides. |
| form | Solid |
| color | White to Off-White |
| Water Solubility | Soluble in water at 3.4mg/ml. |
| Merck | 14577 |
| JECFA Number | 85 |
| BRN | 7507120 |
| CAS DataBase Reference | 61336-70-7(CAS DataBase Reference) |
| EPA Substance Registry System | Amoxicillin trihydrate (61336-70-7) |
| Hazard Codes | Xn,Xi |
| Risk Statements | 42/43 |
| Safety Statements | 36-36/37-22 |
| WGK Germany | 2 |
| RTECS | XH8300000 |
| HS Code | 29411000 |
| Toxicity | LD50 ipr-rat: 2870 mg/kg KSRNAM 7,3040,73 |
Product Application of Amoxicillin Trihydrate CAS#61336-70-7
Amoxicillin is a semi-synthetic broad-spectrum penicillin antibiotic, and its antibacterial spectrum, therapeutic effects, and applications are consistent with those of ampicillin. It is widely used in pharmaceutical formulations for the treatment of susceptible bacterial infections.
Application: This product falls under the classification of pharmacologically active substances and maximum residue limit (MRL) standards for foods of animal origin in accordance with Commission Regulation (EU) No 37/2010. As a semi-synthetic broad-spectrum penicillin, it offers the same antibacterial spectrum, efficacy, and application profile as ampicillin.