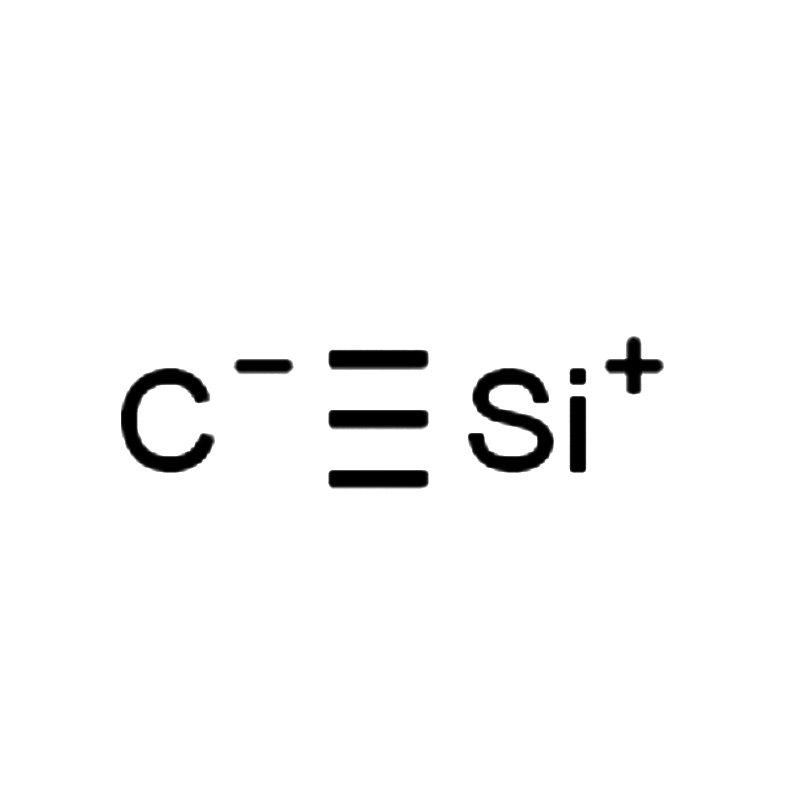Silicon carbide CAS#409-21-2
High Hardness & Wear Resistance – Extremely hard material with excellent abrasion resistance.
Corrosion & Slag Resistance – Performs well against molten slag and harsh environments.
Thermal Shock Resistance – Withstands rapid temperature changes without cracking.
High Conductivity & Multiple Structures – Offers good electrical conductivity and various polytypes for versatile applications.
Silicon Carbide CAS#409-21-2
Silicon carbide is a hard material with strong covalent bonding, mainly produced through the carbothermal reduction of silica. It is manufactured by heating a mixture of silica sand and petroleum coke around electrodes in an electric resistance furnace at temperatures above 2200 °C. Depending on the specific reaction conditions, the product may form as a fine powder or as a bonded mass that must be crushed and milled into a usable feedstock.
This material shows excellent resistance to abrasion and to corrosion by molten slag, along with outstanding thermal shock resistance. As a carbide, it is prone to oxidation, and it also exhibits relatively high electrical conductivity.
Hundreds of different silicon carbide crystal structures (polytypes) have been identified, each with distinct stacking arrangements of silicon and carbon atoms. The simplest form is the cubic diamond-like structure known as β-SiC, while other forms are hexagonal or rhombohedral and are collectively referred to as α-SiC.
Silicon carbide Chemical Properties
| Melting point | 2700 °C (lit.) |
| bulk density | 0.069g/cm3 |
| density | 3.22 g/mL at 25 °C (lit.) |
| refractive index | 2.65 |
| solubility | Soluble in molten sodium hydroxide, potassium hydroxide and in molten iron. |
| form | nanopowder |
| color | Green |
| Specific Gravity | 3.22 |
| Resistivity | 107–200 (ρ/μΩ.cm) |
| Water Solubility | Soluble in molten alkalis (NaOH, KOH) and molten iron. Insoluble in water. |
| Hydrolytic Sensitivity | 1: no significant reaction with aqueous systems |
| Crystal Structure | Cubic, Sphalerite Structure - Space Group F(-4)3m |
| Merck | 148,492 |
| Exposure limits | ACGIH: TWA 10 mg/m3; TWA 3 mg/m3; TWA 0.1 fiber/cm3 |
| OSHA: TWA 15 mg/m3; TWA 5 mg/m3 | |
| NIOSH: TWA 10 mg/m3; TWA 5 mg/m3 | |
| Stability: | Stability |
| Cosmetics Ingredients Functions | ABRASIVE |
| InChIKey | HBMJWWWQQXIZIP-UHFFFAOYSA-N |
| Knoop Microhardness | 3000, at 25°C |
| Shear Modulus | 180 GPa, Calculated |
| Modulus of Elasticity | 459 - 476 GPa |
| Poissons Ratio | 0.14, sintered |
| NIST Chemistry Reference | Silicon monocarbide(409-21-2) |
| IARC | 2A (Vol. 111) 2017 |
| EPA Substance Registry System | Silicon carbide (409-21-2) |
Safety Information
| Hazard Codes | Xi |
| Risk Statements | 36/37/38 |
| Safety Statements | 26-36 |
| OEB | B |
| OEL | TWA: 10 mg/m3 (total) |
| WGK Germany | 3 |
| RTECS | VW0450000 |
| TSCA | Yes |
| HS Code | 28492000 |
| Hazardous Substances Data | 409-21-2(Hazardous Substances Data) |
Product Application of Silicon Carbide CAS#409-21-2
Silicon carbide is one of the few entirely synthetic minerals utilized in refractory applications. The common bonding systems include:
Oxide-bonded (S102, Al2O3, SiO2, or silicate glass)
Silicon oxynitride (Si2ON2)
Silicon nitride (Si3N4)
Among these four bonding types, the first three produce permeable products. When such masonry systems fail due to chemical degradation, it is generally the bond that is attacked. Consequently, permeable units, which allow corrosive agents to penetrate the material, are damaged more quickly.
In contrast, “self-bonded” silicon carbide—where silicon carbide bonds directly to silicon carbide—produces an impermeable material, with corrosion limited to the surface.
Self-bonded silicon carbide can be produced through two methods: reaction bonding or sintering. Both methods yield impermeable units with comparable chemical resistance, although their physical properties are not identical.