LITHIUM METASILICATE CAS#10102-24-6
High Purity White Powder Form: Available as a white powder or orthorhombic needle crystals, ensuring consistent quality and ease of handling.
Defined Thermal Properties: Features a clear enthalpy of fusion (28.00 kJ/mol), supporting stable and predictable high-temperature performance.
Reliable Manufacturing Process: Produced by fusing lithium carbonate with silicon dioxide (SiO₂), ensuring controlled composition and quality.
Effective Fluxing Agent: Widely used as a flux in glazes and ceramic enamels, improving melting behavior and surface finish in ceramic applications.
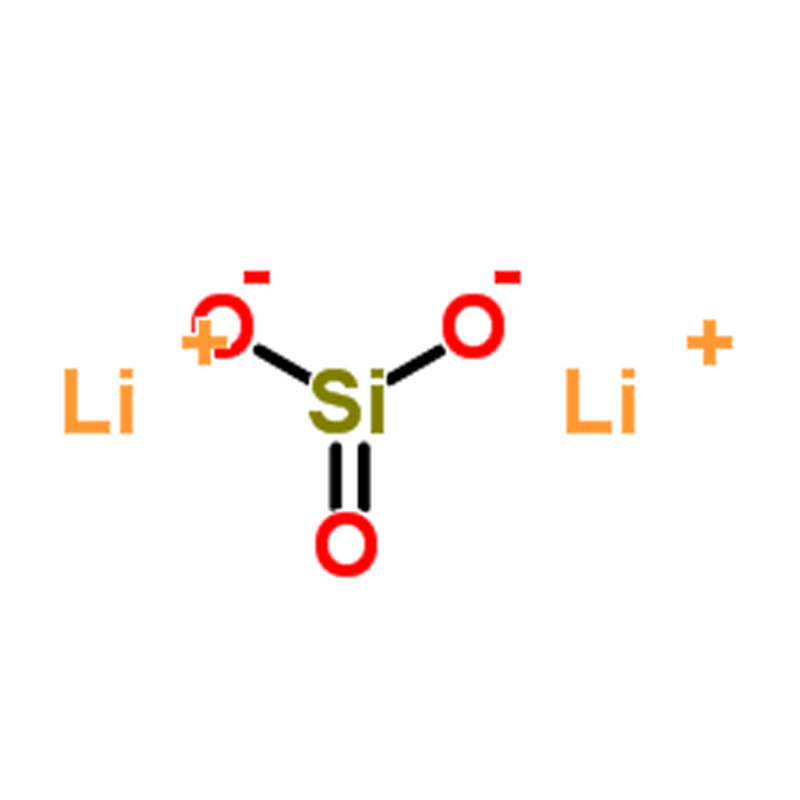
Lithium Metasilicate (CAS#10102-24-6)
Lithium metasilicate appears as a white powder or as orthorhombic needle-shaped crystals. It has an enthalpy of fusion of 28.00 kJ/mol.
The compound is produced by fusing lithium carbonate with silicon dioxide (SiO₂). It is primarily used as a flux in ceramic glazes and enamel formulations, where it helps improve melting characteristics and surface quality.
LITHIUM METASILICATE Chemical Properties
| Melting point | 1204°C |
| density | 2,52 g/cm3 |
| refractive index | 1.584 |
| solubility | insoluble in cold H2O; reacts with dilute acid |
| form | Powder |
| color | white |
| Specific Gravity | 2.52 |
| Odor | Odorless |
| Water Solubility | insoluble cold H2O, decomposes in boiling H2O [MER06] |
| Hydrolytic Sensitivity | 2: reacts with aqueous acid |
| InChI | InChI=1S/2Li.O3Si/c;;1-4(2)3/q2*+1;-2 |
| InChIKey | PAZHGORSDKKUPI-UHFFFAOYSA-N |
| SMILES | [Si]([O-])([O-])=O.[Li+].[Li+] |
| CAS DataBase Reference | 10102-24-6(CAS DataBase Reference) |
| EPA Substance Registry System | Dilithium metasilicate (10102-24-6) |
| Risk Statements | 36/37/38 |
| Safety Statements | 26-36/37/39 |
| WGK Germany | WGK 3 |
| TSCA | TSCA listed |
| Storage Class | 11 - Combustible Solids |
| Hazard Classifications | Eye Irrit. 2 |
| STOT SE 3 |
Product Application of Lithium Metasilicate (CAS#10102-24-6)
Lithium metasilicate is an inorganic compound characterized by strong resistance to heat and acids. It is utilized as an additive in zirconium oxide membranes employed in membrane separation technologies.
The material demonstrates high transport capacity for water vapor and sodium carbonate, contributing to its functional performance in separation systems. Owing to these properties, it has also been explored for use in clinical environments, where it has exhibited biological activity and clinical significance.