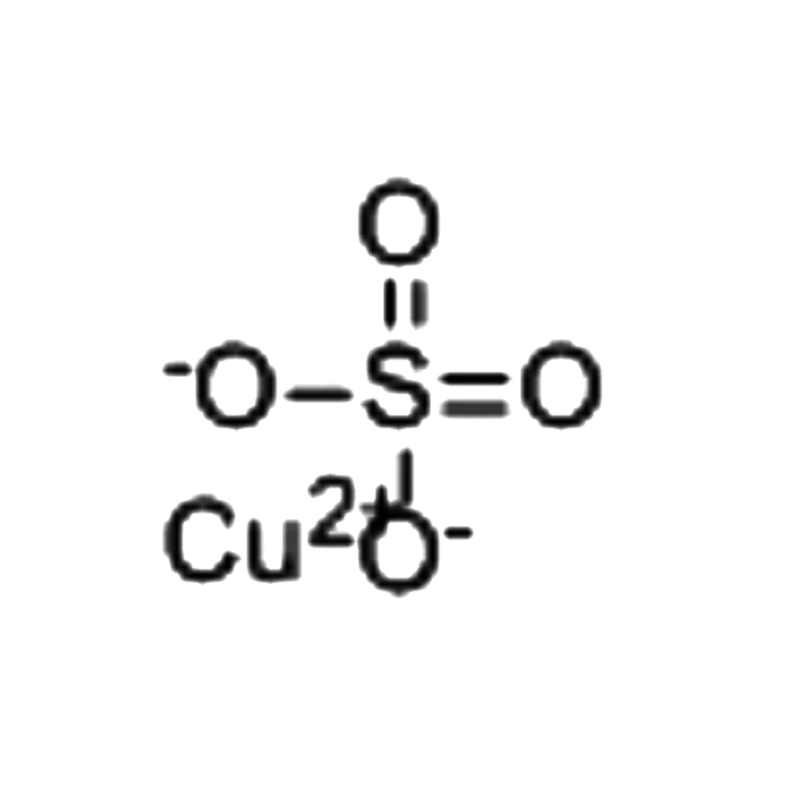
Copper(II) sulfate CAS#7758-98-7
High Water Solubility – Copper sulfate dissolves easily in water, forming a bright blue solution.
Versatile Physical Forms – Available as greenish-white crystalline solid, blue powder, granules, or ultramarine crystalline solid.
Thermal Stability – Pentahydrate decomposes at high temperatures (above 110℃), while anhydrous copper sulfate decomposes to CuO at 650℃.
Safe Handling Profile – Rated low in flammability (0) and reactivity (0), with moderate health hazard (2) according to NFPA-704.
Copper(II) Sulfate CAS#7758-98-7
Copper sulfate appears as a greenish-white crystalline solid, while its pentahydrate form exists as blue powder, granules, or ultramarine crystalline solid. Its molecular weight is 159.60 for the anhydrous form and 249.70 for the pentahydrate. The pentahydrate boils at 150℃ with 25H₂O, and its melting point is around 200℃ (decomposes). The pentahydrate decomposes at approximately 110℃ with 24H₂O, and anhydrous copper sulfate decomposes to CuO at 650℃.
Hazard Identification (according to the NFPA-704 M Rating System): Health 2, Flammability 0, Reactivity 0.
Copper sulfate is highly soluble in water, producing a bright blue solution.
Copper(II) sulfate Chemical Properties
| Melting point | 200 °C (dec.)(lit.) |
| bulk density | 800kg/m3 |
| density | 3.603 g/mL at 25 °C(lit.) |
| vapor pressure | 7.3 mm Hg ( 25 °C) |
| storage temp. | Store at +5°C to +30°C. |
| solubility | H2O: soluble |
| form | powder |
| color | Slightly greenish to gray |
| Specific Gravity | 3.603 |
| Flame Color | Bluish-green |
| Odor | odorless |
| PH | 3.5-4.5 (50g/l, H2O, 20℃) |
| PH Range | 3.7 - 4.5 |
| Water Solubility | 203 g/L (20 ºC) |
| Sensitive | Hygroscopic |
| Merck | 142,653 |
| Exposure limits | ACGIH: TWA 1 mg/m3 |
| NIOSH: IDLH 100 mg/m3; TWA 1 mg/m3 | |
| Dielectric constant | 10.3(0.0℃) |
| Stability: | hygroscopic |
| Cosmetics Ingredients Functions | SKIN CONDITIONING |
| InChIKey | ARUVKPQLZAKDPS-UHFFFAOYSA-L |
| CAS DataBase Reference | 7758-98-7(CAS DataBase Reference) |
| NIST Chemistry Reference | Cupric sulfate(7758-98-7) |
| EPA Substance Registry System | Cupric sulfate (7758-98-7) |
Safety Information
| Hazard Codes | Xn,N,Xi |
| Risk Statements | 36/38-50/53-22-51/53-36/37/38 |
| Safety Statements | 24/25-36-60-61-22-26 |
| RIDADR | UN 3288 6.1/PG 3 |
| WGK Germany | 2 |
| RTECS | GL8800000 |
| F | 3 |
| TSCA | Yes |
| HS Code | 2833 25 00 |
| HazardClass | 6.1 |
| PackingGroup | III |
| Hazardous Substances Data | 7758-98-7(Hazardous Substances Data) |
| Toxicity | LD50 orally in Rabbit: 481 mg/kg |
Product Application of Copper(II) Sulfate CAS#7758-98-7
Copper sulfate, also known as blue vitriol, is produced by the reaction of sulfuric acid with elemental copper. Its bright-blue crystals are soluble in both water and alcohol. When combined with ammonia, copper sulfate has been used in liquid filtration systems. A common application involves mixing it with potassium bromide to produce copper bromide bleach for image intensification and toning. Additionally, some photographers have used copper sulfate as a restrainer in ferrous sulfate developers employed in the collodion photographic process.