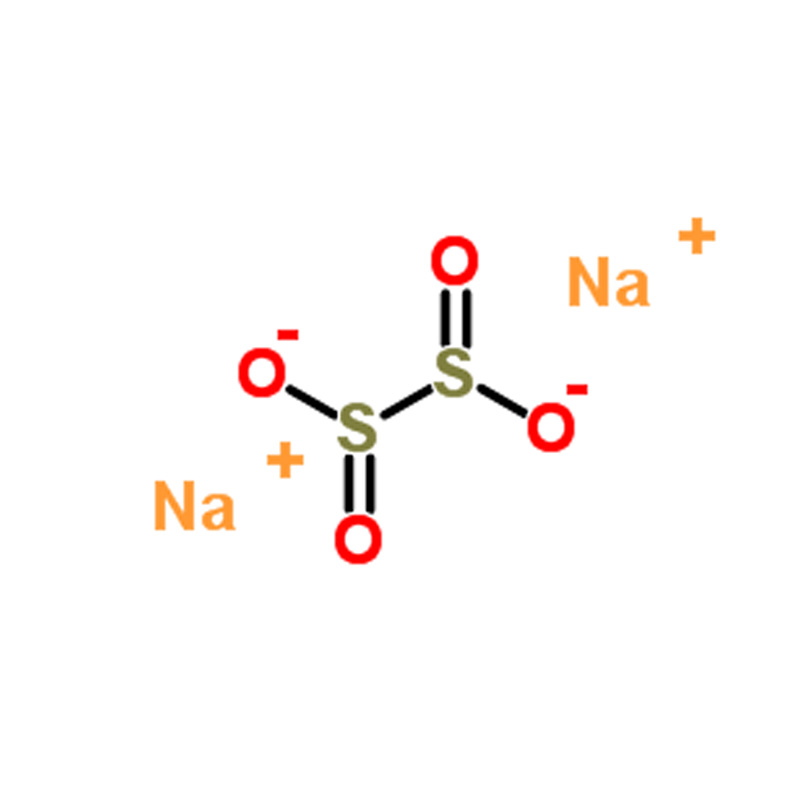
Sodium dithionite CAS#7775-14-6
Strong Reducing Performance
Exhibits powerful reducing ability and high oxygen reactivity, enabling efficient bleaching, decolorization, and reduction processes.
Broad Industrial Applicability
Widely used across textiles, pulp and paper, water treatment, photography, polymers, leather, food and beverage processing, and chemical manufacturing.
Effective Oxygen Scavenging
Performs efficiently as an oxygen remover in boilers and industrial systems, helping to prevent oxidation and corrosion.
High Process Efficiency
Rapid reaction and conversion characteristics support fast processing, effective stain removal, and reliable performance in conservation, environmental remediation, and metal recovery applications.
Product Description of Sodium Dithionite CAS#7775-14-6
Sodium dithionite, also known as sodium hydrosulfite, sodium hydrosulphite, sodium sulfoxylate, or sulfoxylate, is a strong reducing agent. It is unstable under physiological conditions, with decomposition accelerating in acidic environments. When exposed to moisture, it is oxidized to hydrogen sulfite, sulfite, and hydrogen sulfate, and under strongly acidic conditions may release sulfur dioxide. In anaerobic environments, hydrogen sulfite and thiosulfate can form. After ingestion, hydrogen sulfite may be absorbed, rapidly metabolized, and mainly excreted as sulfate in urine.
Owing to its strong reducing ability and high reactivity with oxygen, sodium dithionite is widely used across various industries. Its applications include textile dyeing, pulp and paper bleaching to remove yellowing from cellulose-based materials, oxygen scavenging in boilers, conservation for removing iron stains from cultural artifacts, and water treatment to control iron-related discoloration during bleaching. It is also used in photographic processing, clay and ceramic production, wine making, leather processing, food and beverage industries, polymer manufacturing, cleaning formulations, gas purification, environmental remediation, metal recovery, and chemical processing.
Sodium dithionite Chemical Properties
| Melting point | 300 °C |
| Boiling point | 1390°C |
| bulk density | 1250kg/m3 |
| density | 2.13 |
| Fp | >100°C |
| storage temp. | Store at +5°C to +30°C. |
| solubility | 250 g/L (20°C) |
| form | Powder/Solid |
| color | White |
| Odor | None or slight scent of sulfur dioxide |
| PH | 5.5-8.5 (50g/l, H2O, 20℃) |
| Water Solubility | 250 g/L (20 ºC) |
| Sensitive | Moisture Sensitive |
| Merck | 148,626 |
| Stability: | Stable, but air sensitive. Incompatible with strong acids, strong oxidizing agents, water, moisture. |
| Cosmetics Ingredients Functions | VISCOSITY CONTROLLING |
| REDUCING | |
| InChI | 1S/2Na.H2O4S2/c;;1-5(2)6(3)4/h;;(H,1,2)(H,3,4)/q2*+1;/p-2 |
| InChIKey | JVBXVOWTABLYPX-UHFFFAOYSA-L |
| SMILES | [Na+].[Na+].[O-]S(=O)S([O-])=O |
| LogP | -2.756 (est) |
| CAS DataBase Reference | 7775-14-6(CAS DataBase Reference) |
| EPA Substance Registry System | Sodium hydrosulfite (7775-14-6) |
Safety Information
| Hazard Codes | Xn |
| Risk Statements | 7-22-31 |
| Safety Statements | 26-28-43-7/8-43E-28A |
| RIDADR | UN 1384 4.2/PG 2 |
| WGK Germany | 1 |
| F | 1月10日 |
| Autoignition Temperature | >200 °C |
| TSCA | TSCA listed |
| HazardClass | 4.2 |
| PackingGroup | II |
| HS Code | 28311010 |
| Storage Class | 4.2 - Pyrophoric and self-heating hazardous materials |
| Hazard Classifications | Acute Tox. 4 Oral |
| Eye Irrit. 2 | |
| Self-heat. 1 | |
| Hazardous Substances Data | 7775-14-6(Hazardous Substances Data) |
| Toxicity | LD50 orally in Rabbit: 2500 mg/kg |
Product Application of Sodium Dithionite CAS#7775-14-6
Sodium dithionite, also known as sodium hydrosulfite, is widely used in aqueous systems as a reducing agent, sulfonating agent, chelating agent, and decolorizing agent in organic reactions. It is applied in water treatment, gas purification, cleaning formulations, leather processing, polymer production, photography, and many other industrial fields. In chemically enhanced oil recovery, it is used to stabilize polyacrylamide polymers against radical degradation in the presence of iron. Additionally, sodium dithionite plays an important role in soil chemistry for determining iron content.
Factory and Equipment Show