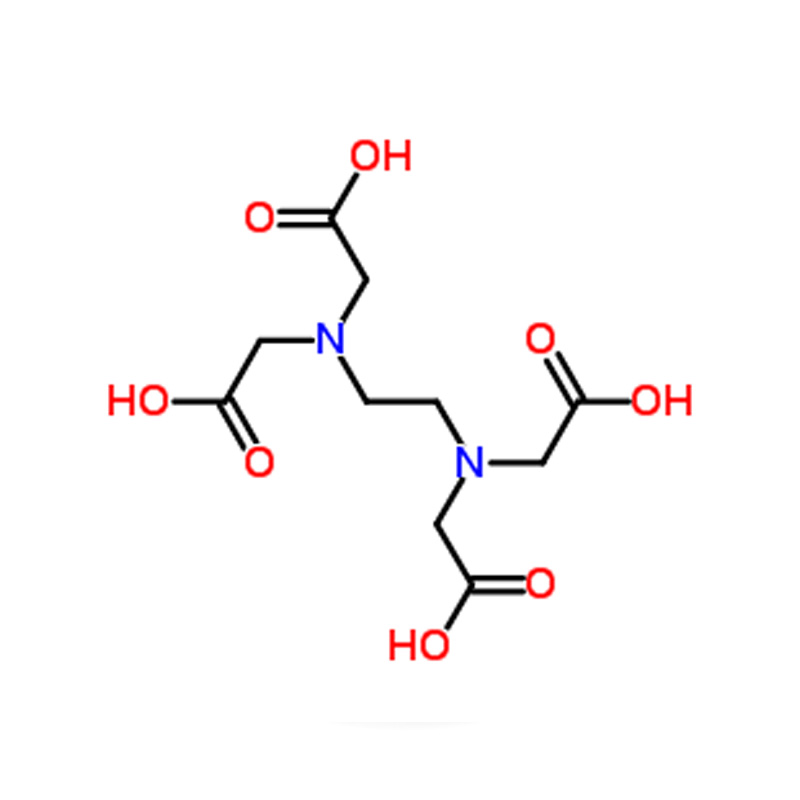
Ethylenediaminetetraacetic acid CAS#60-00-4
Strong Chelating Ability: EDTA can bind to metal ions at six different sites, forming highly stable complexes.
Multi-Functional Binding: It contains two nitrogen and four oxygen atoms, allowing versatile coordination with various metals.
Effective Metal Deactivation: EDTA’s strong grasp on metal ions can neutralize or deactivate them efficiently.
Reliable Stability: The hexadentate chelation structure ensures that the formed metal complexes remain stable under diverse conditions.
Product Description of Ethylenediaminetetraacetic Acid CAS# 60-00-4
Ethylenediaminetetraacetic Acid (EDTA) is a widely used polydentate ligand. In solution, its hydrogen atoms are easily removed, forming the anionic EDTA⁴⁻. In this anionic form, EDTA possesses six binding atoms—two nitrogen and four oxygen atoms.
EDTA can attach to a metal ion at all six binding sites, effectively surrounding the ion and forming a highly stable complex. Its strong hold on metal ions is often compared to a crab or lobster gripping an object with its claw, which is the origin of the term "chelation." This hexadentate binding makes EDTA a highly effective chelating agent, capable of deactivating metals at up to six sites simultaneously.
Ethylenediaminetetraacetic acid Chemical Properties
| Melting point | 250 °C (dec.) (lit.) |
| Boiling point | 434.18°C (rough estimate) |
| bulk density | 600kg/m3 |
| density | 1.46 g/cm3 at 20 °C |
| vapor pressure | <0.013 hPa (20 °C) |
| refractive index | n20/D 1.363 |
| Fp | >400°C DIN 51758 |
| storage temp. | 2-8°C |
| solubility | 3 M NaOH: 100 mg/mL |
| form | crystalline |
| pka | pKa 2 (Uncertain);10.26 (Uncertain) |
| color | White to almost white |
| PH | 2.5 (10g/l, H2O, 23℃)(slurry) |
| Odor | Odorless |
| PH Range | 2.5 at 10 g/l at 23 °C |
| Water Solubility | 0.5 g/L (25 ºC) |
| λmax | λ: 280 nm Amax: ≤0.25 |
| Decomposition | 240 °C |
| Merck | 143517 |
| BRN | 1716295 |
| Stability: | Stable. Incompatible with copper, copper alloys, nickel, aluminium, strong oxidizing agents, strong bases |
| LogP | -0.836 (est) |
| CAS DataBase Reference | 60-00-4(CAS DataBase Reference) |
| NIST Chemistry Reference | N,N'-1,2-Ethane diylbis-(N-(carboxymethyl)glycine)(60-00-4) |
| EPA Substance Registry System | Ethylenediaminetetraacetic acid (60-00-4) |
Safety Information
| Hazard Codes | Xi |
| Risk Statements | 36-52/53-36/37/38-36/38 |
| Safety Statements | 26-61-37/39-36 |
| RIDADR | UN 3077 9 / PGIII |
| WGK Germany | 2 |
| RTECS | AH4025000 |
| F | 3 |
| Autoignition Temperature | >200 °C |
| TSCA | Yes |
| HS Code | 2922 49 85 |
| Hazardous Substances Data | 60-00-4(Hazardous Substances Data) |
| Toxicity | LD50 orally in Rabbit: 2580 mg/kg |
Product Application of Ethylenediaminetetraacetic Acid CAS# 60-00-4
Ethylenediamine-N,N,N’,N’-tetraacetic Acid (EDTA) is a highly effective chelating agent that forms stable complexes with a wide range of metal ions. It is commonly used in the treatment of lead and other heavy metal poisoning in farm animals.