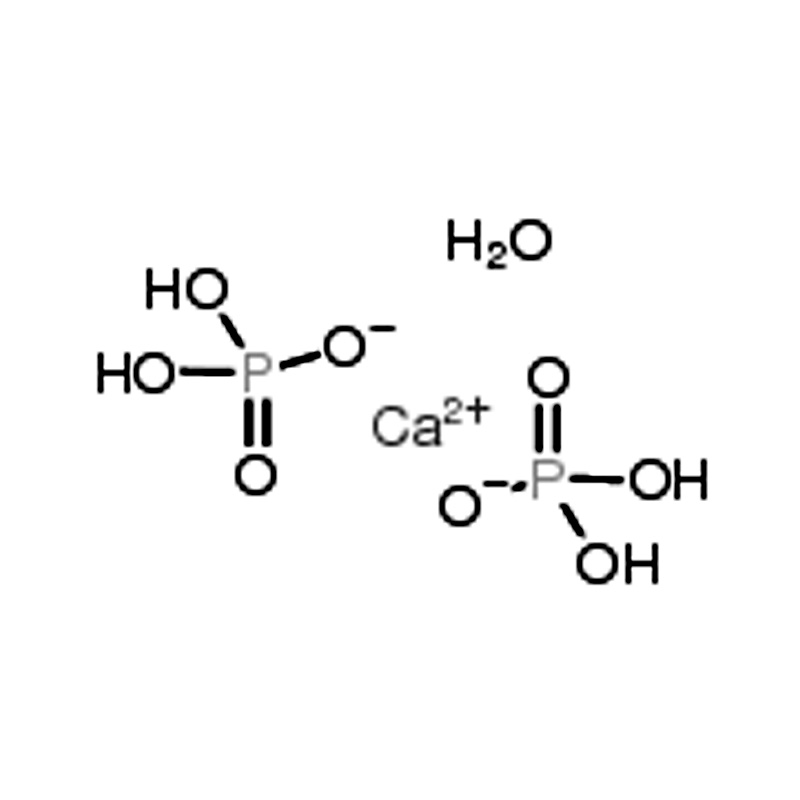
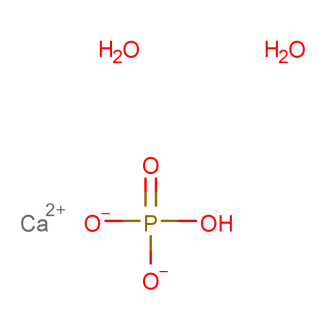
Dual-Industry Versatility – Widely used in both fertilizer production and the food industry as an effective acidulant, meeting diverse industrial and nutritional processing needs.
Excellent Reactivity in Cement Formation – Reacts efficiently with α-tricalcium phosphate in aqueous systems to form dicalcium phosphate dihydrate cement with reliable setting performance.
Ideal for Brushite Cement Applications – Serves as a key raw material in the preparation of brushite cement, supporting biomedical and construction-related formulations.
Stable and Process-Friendly Compound – Provides consistent chemical behavior in aqueous solutions, enabling controlled cement composition and dependable product quality.
Product Details
Product Description of Calcium Phosphate Monobasic (CAS No. 10031-30-8)
Calcium Phosphate Monobasic Monohydrate is widely utilized in fertilizer production and as an acidifying agent in the food processing industry. When dissolved in water and combined with α-tricalcium phosphate, it reacts to form dicalcium phosphate dihydrate cement with reliable setting properties.
In addition, this compound is an important component in the formulation of brushite cement, supporting stable performance in specialized cement and biomedical applications.

| Melting point | 100°C |
| Boiling point | 203°C |
| density | 2.220^1^6 |
| Fp | 203°C |
| solubility | slightly soluble in H2O; soluble in dilute acid solutions |
| form | Powder |
| color | white |
| Odor | Odorless |
| Water Solubility | moderately soluble H2O; soluble dilute HCl, HNO3, acetic acid [MER06] |
| Merck | 141,693 |
| Dielectric constant | 14.0(Ambient) |
| InChIKey | ZBZJARSYCHAEND-UHFFFAOYSA-L |
| CAS DataBase Reference | 10031-30-8(CAS DataBase Reference) |
| EPA Substance Registry System | Monocalcium phosphate monohydrate (10031-30-8) |
| Hazard Codes | Xi |
| Risk Statements | 36/37/38 |
| Safety Statements | 26-36/37 |
| WGK Germany | 2 |
| RTECS | TB8530000 |
| HS Code | 31031090 |
Product Application of Calcium Phosphate Monobasic (CAS No. 10031-30-8)
Monocalcium phosphate monohydrate is one of the earliest acidic phosphates used as a leavening agent in the baking industry, with its application in baking powders officially patented in 1956. Early production was based on calcined bone materials to obtain a crude phosphate product. This compound is classified as a fast-acting leavening acid, releasing about 60% of its theoretical carbon dioxide during the mixing stage when it reacts with baking soda in dough or batter. Further gas release occurs only after baking begins and the temperature reaches approximately 140 °F.
This behavior results from its reaction with water, where MCP·H₂O partially converts into dicalcium phosphate and phosphoric acid, controlling the timing of gas generation. Due to its rapid reaction rate, monocalcium phosphate monohydrate is rarely used alone. It is commonly added to phosphated flour at levels of 0.25–0.75% of the final product weight and is widely used in cookie formulations. In most applications, it is blended with slower-acting phosphate leavening acids to ensure balanced gas release during mixing, resting, and baking, thereby improving dough structure and final product texture.
Factory and Equipment Show
Leave your messages