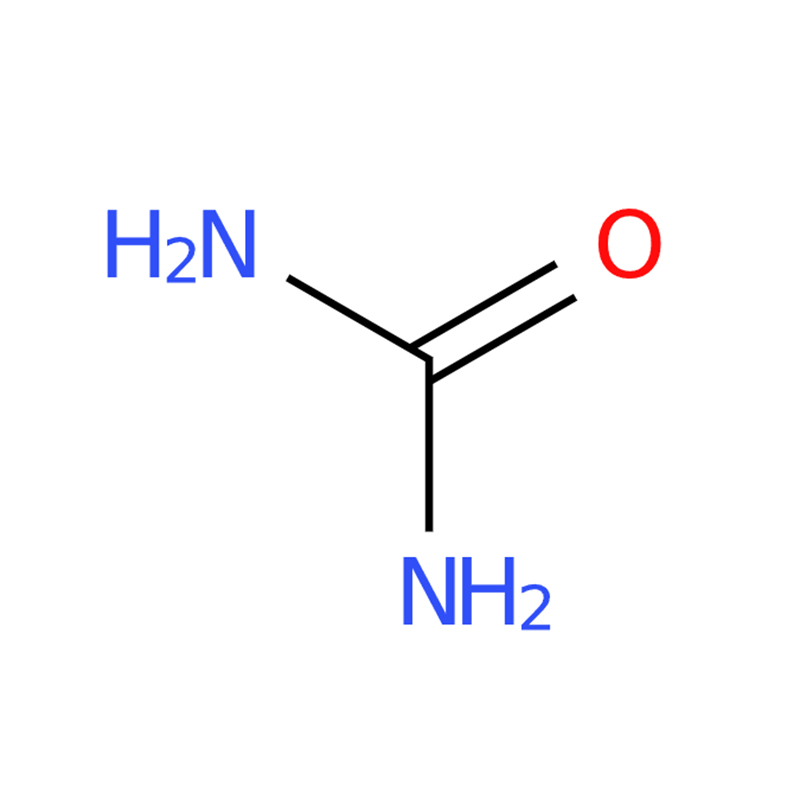
Urea CAS#57-13-6
Strong Hydrogen-Bonding Capability: Urea forms an extensive and stable hydrogen-bond network both in the solid state and in aqueous systems, enhancing its interaction with other molecules.
Excellent Water Solubility: Due to its strong affinity for hydrogen bonding with water, urea dissolves readily, enabling efficient use in aqueous formulations and processing systems.
Chemically Stable and Reactive Structure: The sp²-hybridized carbon and partial double-bond character of the C–N bonds provide structural stability while maintaining useful chemical reactivity.
High Functional Polarity: The relatively basic carbonyl oxygen enhances urea’s molecular polarity, improving compatibility and performance in chemical, agricultural, and industrial applications.
The urea molecule adopts a planar configuration in the crystalline state, whereas in the gas-phase minimum-energy structure, the nitrogen atoms display a pyramidal geometry. In the solid phase, the oxygen atom participates in two N–H···O hydrogen bonds, forming a compact and energetically stable hydrogen-bonding network. This network compromises ideal molecular packing, leading to an open structure composed of ribbon-like arrangements that create square cross-sectional channels.
The carbon atom in urea is sp²-hybridized, which imparts significant double-bond character to the C–N bonds, while the carbonyl oxygen exhibits greater basicity compared to that in compounds such as formaldehyde. The high solubility of urea in water results from its strong ability to form extensive hydrogen bonds with water molecules.
Urea Chemical Properties
Melting point | 132-135 °C(lit.) |
Boiling point | 332.48°C (estimate) |
density | 1.335 g/mL at 25 °C(lit.) |
vapor pressure | <0.1 hPa (20 °C) |
refractive index | n20/D 1.40 |
storage temp | 2-8°C |
solubility | H2O: 8 M at 20 °C |
form | powder |
pka | 0.10(at 25℃) |
color | white |
Specific Gravity | 1.335 |
Odor | almost odorless |
PH | 8.0-10.0 (20℃, 8M in H2O) |
Water Solubility | 1080 g/L (20 ºC) |
λmax | λ: 260 nm Amax: 0.03 |
Merck | 14,9867 |
BRN | 635724 |
Dielectric constant | 3.5(Ambient) |
Stability | Substances to be avoided include strong oxidizing agents. Protect from moisture. |
InChIKey | XSQUKJJJFZCRTK-UHFFFAOYSA-N |
LogP | -1.660 (est) |
CAS DataBase Reference | 57-13-6(CAS DataBase Reference) |
NIST Chemistry Reference | Urea(57-13-6) |
EPA Substance Registry System | Urea (57-13-6) |
Safety Information
Hazard Codes | Xn,Xi |
Risk Statements | 36/37/38-40-38 |
Safety Statements | 26-36-24/25-37 |
RIDADR | Not regulated |
WGK Germany | 1 |
RTECS | YR6250000 |
TSCA | Yes |
HS Code | 31021010 |
Hazardous Substances Data | 57-13-6(Hazardous Substances Data) |
Toxicity | LD50 orally in Rabbit: 8471 mg/kg LD50 dermal Rat 8200 mg/kg |
Urea serves as a physiological regulator of nitrogen excretion in mammals. It is synthesized in the liver as a byproduct of protein metabolism and eliminated from the body through urine. Urea is also naturally present in the skin, where it acts as an emollient and exhibits diuretic properties.
In laboratory and biochemical applications, urea is commonly used to denature proteins and as a mild solubilizing agent for insoluble or denatured proteins. It is especially effective in the renaturation of proteins from samples previously denatured with 6 M guanidine chloride, including inclusion bodies. Additionally, urea can be used in combination with guanidine hydrochloride and dithiothreitol (DTT) to refold denatured proteins into their native or biologically active conformations.