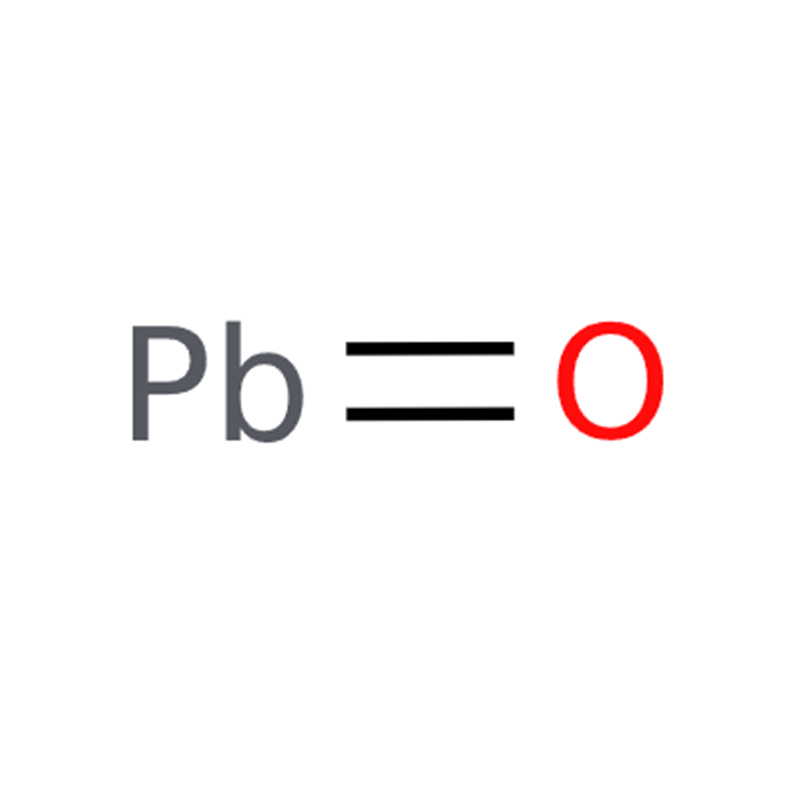Product Application of Lead Monoxide CAS#1317-36-8
Lead monoxide is used in ointments and plasters, and in the preparation of lead subacetate solutions. It is also applied in pottery glazing, as well as a glass flux for decorative painting on porcelain and glass, and in the production of lead glass and varnishes.
In combination with glycerol, it can be used as a metal cement. Additionally, it is employed to create iridescent colors on brass and bronze, and for coloring sulfur-containing materials such as hair, nails, wool, and horn.
Other applications include the manufacture of artificial tortoise shell and horn, use as a pigment in rubber, production of boiled linseed oil, and in the assay of gold and silver ores.