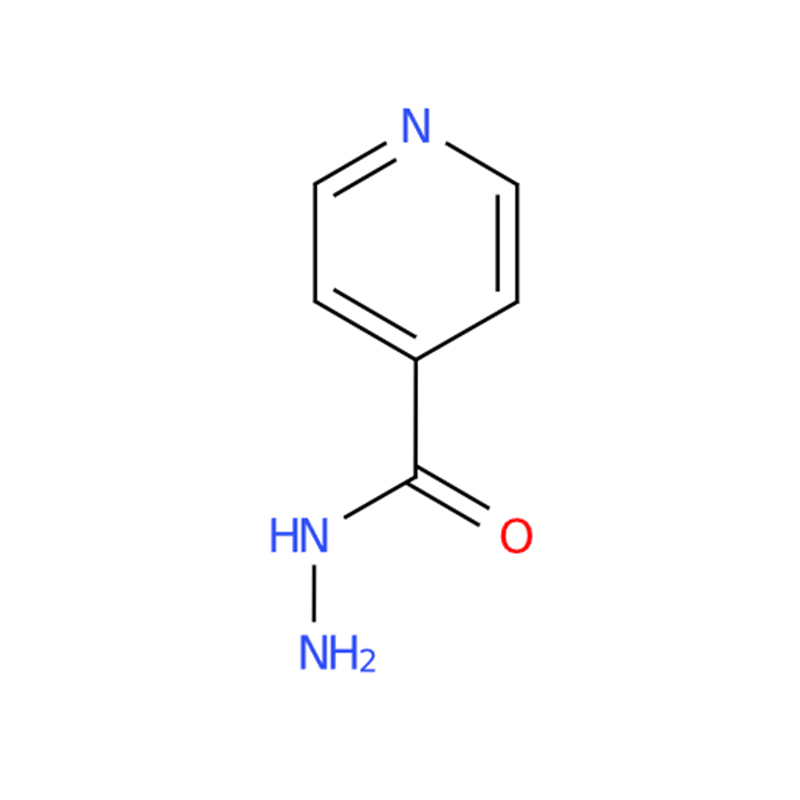
Isoniazid CAS#54-85-3
Excellent Stability and Water Solubility: Isoniazid is chemically stable and readily soluble in water, supporting convenient formulation and broad application use.
Strong Reducing Properties: The hydrazine group provides excellent reducing capability, enabling reactions such as the conversion of dissolved oxygen to hydrogen peroxide under alkaline conditions.
Versatile Chemical Reactivity: Its active amino group (-NH₂) allows efficient reactions with carbonyl compounds, carboxylic acid derivatives, and sulfonyl chlorides, making it a valuable intermediate for derivative synthesis.
Suitable for Analytical and Luminescent Systems: It can participate in gold nanoparticle-catalyzed luminol luminescent systems, offering potential advantages in analytical and detection applications.
Product Description of Isoniazid CAS#54-85-3
Isoniazid appears as a colorless crystal or white crystalline powder with a slightly bitter taste. It is chemically stable and readily soluble in water, making it suitable for a wide range of pharmaceutical and chemical applications.
The hydrazine group in isoniazid exhibits strong reducing properties. Under alkaline conditions, it can slowly reduce dissolved oxygen in water to hydrogen peroxide. This reaction can be catalyzed by gold nanoparticles, forming a luminol-based luminescent system with potential analytical applications.
In addition, the amino group (-NH₂) attached to the hydrazine moiety provides versatile chemical reactivity. It can react with active carbonyl compounds to form Schiff bases, and also undergo reactions with carboxylic acid derivatives or sulfonyl chlorides to produce acylated isoniazid or sulfonyl isoniazid derivatives.
Parameters
| Melting point | 171-173 °C (lit.) |
| Boiling point | 251.97°C (rough estimate) |
| density | 1.2620 (rough estimate) |
| bulk density | 800kg/m3 |
| refractive index | 1.6910 (estimate) |
| Fp | >250°C |
| storage temp. | 2-8°C |
| solubility | 125g/l |
| form | Crystals or Crystalline Powder |
| pka | pKa 2.00/3.60/10.8(H2O) (Uncertain) |
| color | White or colorless |
| PH | 6-8 (50g/l, H2O, 20℃) |
| Odor | Odorless |
| PH Range | 5.5 - 6.5 at 10 g/l at 25 °C |
| Water Solubility | 14 g/100 mL (25 ºC) |
| Sensitive | Air Sensitive |
| Merck | 145186 |
| BRN | 119374 |
| BCS Class | 1,3 |
| Stability: | Stability Stable, but may be air or light sensitive. Combustible. Incompatible with strong oxidizing agents, chloral, aldehydes, iodine, ferric salts, hypochlorites. |
| InChIKey | QRXWMOHMRWLFEY-UHFFFAOYSA-N |
| CAS DataBase Reference | 54-85-3(CAS DataBase Reference) |
| NIST Chemistry Reference | Isoniazid(54-85-3) |
| IARC | 3 (Vol. 4, Sup 7) 1987 |
| EPA Substance Registry System | Isoniazid (54-85-3) |
Safety Information
| Hazard Codes | Xn |
| Risk Statements | 22-38-40-36/37/38 |
| Safety Statements | 37-36/37/39-26 |
| RIDADR | 2811 |
| WGK Germany | 3 |
| RTECS | NS1751850 |
| TSCA | Yes |
| PackingGroup | III |
| HS Code | 29333999 |
| Hazardous Substances Data | 54-85-3(Hazardous Substances Data) |
| Toxicity | LD50 in mice (mg/kg): 151 i.p., 149 i.v. (Jenney, Pfeiffer) |
Product Application of Isoniazid CAS#54-85-3
Isoniazid is widely used as a first-line anti-tuberculosis drug, valued for its high efficacy, low toxicity, and cost-effectiveness. In addition to its pharmaceutical use, it is also applied as an electroplating additive and pharmaceutical intermediate.
In chemical synthesis, the condensation of isoniazid with formaldehyde sodium bisulfite can produce isoniazid methanesulfonic acid (CAS#13447-95-5). It can also react with formaldehyde to form di-isoniazid. This condensation process is relatively straightforward, typically carried out by adding formaldehyde to an aqueous isoniazid solution and reacting at approximately 65°C for 30 minutes.
The condensation of isoniazid with vanillic acid is also performed in aqueous solution at around 95°C, yielding isoniazid hydrazone. These derivatives also possess anti-tuberculosis activity. Among them, isoniazid itself demonstrates the strongest efficacy, while isoniazid hydrazone shows lower activity against Mycobacterium tuberculosis but reduced neurotoxicity and hepatotoxicity compared with isoniazid.
For biochemical research and clinical applications, isoniazid remains one of the most important anti-tuberculosis agents. It exhibits high selectivity and strong antibacterial activity against tuberculosis bacteria. In vitro, concentrations as low as 0.025–0.05 mg/L can inhibit bacterial growth, while higher concentrations exert a bactericidal effect on actively reproducing bacteria.