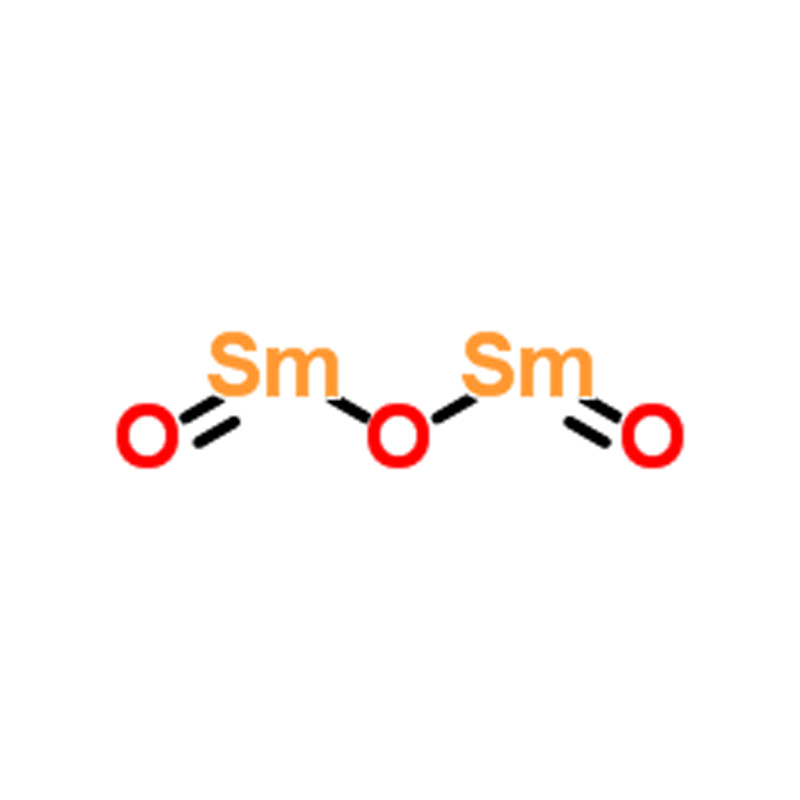
Samarium oxide CAS#12060-58-1
Excellent Thermal Stability and Insolubility: Highly insoluble and thermally stable, making it suitable for demanding high-temperature applications.
Broad High-Tech Application Range: Widely used in glass, ceramics, phosphors, lasers, thermoelectric devices, and infrared-absorbing optical materials.
Effective Catalytic Performance: Acts as an efficient catalyst and chemical reagent, particularly for the dehydration of acyclic primary alcohols into aldehydes and ketones.
Nuclear and Advanced Functional Utility: Functions as a neutron absorber in nuclear reactor control rods and enhances infrared radiation absorption when incorporated into ceramics and specialty glass.
Samarium Oxide (CAS#12060-58-1)
Samarium oxide is a highly insoluble and thermally stable samarium source with specialized applications in glass, ceramics, phosphors, lasers, and thermoelectric devices.
Samarium and its compounds are also widely used as catalysts and chemical reagents, particularly for promoting the dehydration of acyclic primary alcohols to produce aldehydes and ketones. In its common oxidized form, samarium can be incorporated into ceramics and optical or infrared-absorbing glass to enhance infrared radiation absorption. It is additionally utilized as a neutron absorber in control rods for nuclear power reactors.
Furthermore, samarium oxide serves as a precursor for the preparation of other samarium salts through dissolution in mineral acids followed by evaporation and crystallization. It can be produced by burning samarium metal in air or oxygen above 150 °C, or by the thermal decomposition of samarium carbonate, nitrate, oxalate, or sulfate.
Samarium oxide Chemical Properties
| Melting point | 2325 °C |
| Boiling point | 4118.00°C |
| Bulk density | 850kg/m3 |
| Density | 8.35 g/mL at 25 °C(lit.) |
| Refractive index | 1.97 |
| Storage temp | no restrictions. |
| Form | nanopowder |
| Color | Violet |
| Specific Gravity | 8.347 |
| PH | 8.0 (10g/l, H2O, 25℃)(slurry) |
| Water Solubility | Insoluble in water. |
| Crystal Structure | Cubic |
| Crystal system | Monoclinic |
| Merck | 148347 |
| Space group | C2/m |
| Lattice constant | a/nmb/nmc/nmα/oβ/oγ/oV/nm31.41770.36330.88479099.96900.4488 |
| InChI | InChI=1S/3O.2Sm |
| InChIKey | PRCWVHVINXAFRG-UHFFFAOYSA-N |
| SMILES | O([Sm]=O)[Sm]=O |
| CAS DataBase Reference | 12060-58-1(CAS DataBase Reference) |
| EPA Substance Registry System | Samarium oxide (Sm2O3) (12060-58-1) |
Safety Information
| Safety Statements | 22-24/25 |
| WGK Germany | 2 |
| RTECS | VP3153000 |
| F | 3-10-34 |
| TSCA | TSCA listed |
| HS Code | 28469019 |
| Storage Class | 10 - Combustible liquids |
| Toxicity | LD50 orally in Rabbit: > 5000 mg/kg |
Product Application of Samarium Oxide (CAS#12060-58-1)
Samarium oxide, also known as samaria, possesses a high neutron absorption capacity. It is widely applied in glass, phosphors, lasers, and thermoelectric devices. Samarium-treated calcium chloride crystals have been used in laser systems capable of generating highly intense light beams strong enough to burn metal or reflect off the moon.
In addition, samarium oxide is incorporated into optical and infrared-absorbing glass to enhance infrared radiation absorption. It serves as a neutron absorber in control rods for nuclear power reactors. The oxide also functions as a catalyst for the dehydration of acyclic primary alcohols to form aldehydes and ketones. Furthermore, it is used as a precursor in the preparation of other samarium salts.