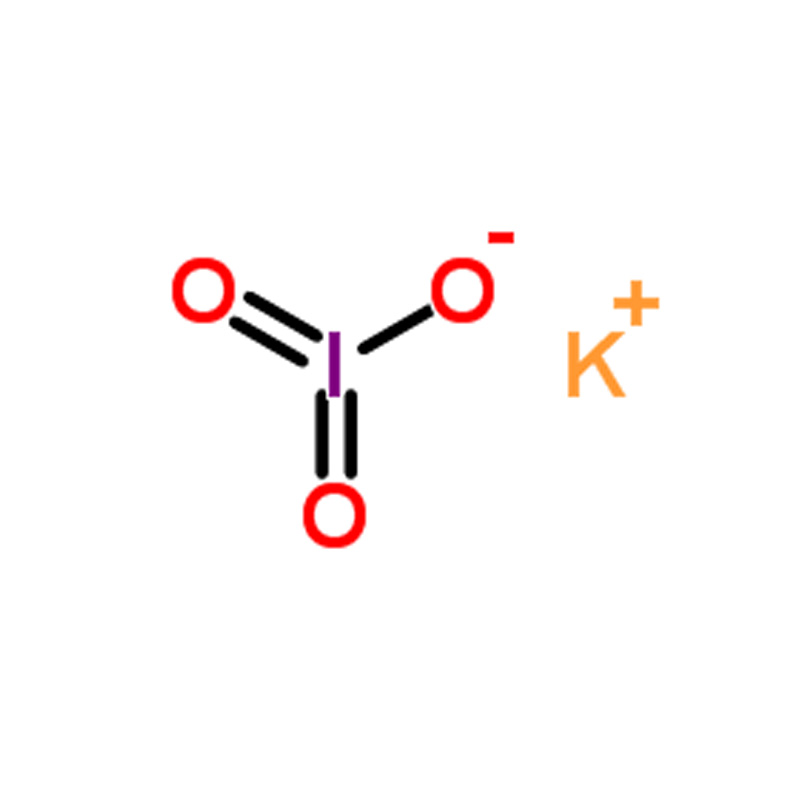
Potassium iodate CAS#7758-05-6
Excellent Chemical Stability: More stable than potassium iodide, especially in hot and humid environments, offering a longer shelf life.
Effective Iodine Source: An iodine-rich salt that provides reliable iodine content for various applications.
Radiation Protection Capability: Acts as a strong blocker of radioactive iodine uptake by the thyroid gland.
Good Solubility and Handling Properties: Soluble in water, dilute acids, and several amine solutions, with stable crystalline form and high thermal stability under normal conditions.
Potassium Iodate CAS No. 7758-05-6
Potassium iodate is an iodine-rich salt with the chemical formula KIO₃. Compared with potassium iodide, it exhibits greater chemical stability, particularly in hot and humid environments where potassium iodide may undergo hydrolysis due to its hygroscopic nature. As a result, potassium iodate offers a longer shelf life. It is also an effective agent for blocking the uptake of radioactive iodine by the thyroid gland.
Potassium iodate appears as colorless monoclinic crystals or a white crystalline powder and is odorless. It is soluble in water, dilute acids, ethylenediamine, ethanolamine, and aqueous potassium iodide solutions, while being slightly soluble in liquid sulfur dioxide and insoluble in alcohols and ammonia. The compound has a monoclinic crystal structure with a density of 3.90 g/cm³. It is stable at normal temperatures and melts at approximately 560 °C with partial decomposition, releasing oxygen. Its solubility in water increases with temperature, being moderately soluble in cold water (4.74 g/100 mL at 0 °C) and highly soluble in boiling water (32.3 g/100 mL at 100 °C).
Potassium iodate Chemical Properties
| Melting point | 560 °C(lit.) |
| density | 3.93 g/mL at 25 °C(lit.) |
| bulk density | 2000kg/m3 |
| vapor pressure | 0-0Pa at 25℃ |
| storage temp. | Store at +5°C to +30°C. |
| solubility | H2O: 0.1 M at 20 °C, clear, colorless |
| form | Powder/Solid |
| pka | 0.047[at 20 ℃] |
| color | White to off-white |
| Odor | at 100.00?%. odorless |
| PH | 6 (50g/l, H2O, 20℃) |
| Water Solubility | Soluble |
| Merck | 147,642 |
| Solubility Product Constant (Ksp) | pKsp: 3.43 |
| Stability: | Stable. Materials to avoid include organics, combustibles, phosphorus, sulfur, carbon, powdered metals, cyanides, hydrides, strong reducing agents, aluminium, peroxides. Explosive when mixed with combustible material. |
| InChIKey | JLKDVMWYMMLWTI-UHFFFAOYSA-M |
| LogP | -1 at 25℃ |
| CAS DataBase Reference | 7758-05-6(CAS DataBase Reference) |
| EPA Substance Registry System | Potassium iodate (7758-05-6) |
Safety Information
| Hazard Codes | O,Xi,Xn,T |
| Risk Statements | 8-36/37/38-22-61-20/21/22 |
| Safety Statements | 26-37/39-17-24/25-23-53-45-36/37/39-22 |
| RIDADR | UN 1479 5.1/PG 2 |
| WGK Germany | 3 |
| RTECS | NN1350000 |
| F | 21 |
| TSCA | TSCA listed |
| HazardClass | 5.1 |
| PackingGroup | II |
| HS Code | 28299080 |
| Hazardous Substances Data | 7758-05-6(Hazardous Substances Data) |
Product Application of Potassium Iodate CAS No. 7758-05-6
Potassium iodate is a relatively strong oxidizing agent and is commonly used in the assay of various pharmaceutical substances, including benzalkonium chloride, cetrimide, hydralazine hydrochloride, potassium iodide, phenylhydrazine hydrochloride, semicarbazide hydrochloride, and related compounds. Under suitable experimental conditions, iodate reacts quantitatively with both iodides and iodine. Notably, iodate titrations can be effectively performed in the presence of saturated organic acids, alcohols, and many other organic substances.
Oxidation–reduction analytical methods involving potassium iodate are typically based on the formation of iodine monochloride (ICl) in a strongly acidic hydrochloric acid medium.