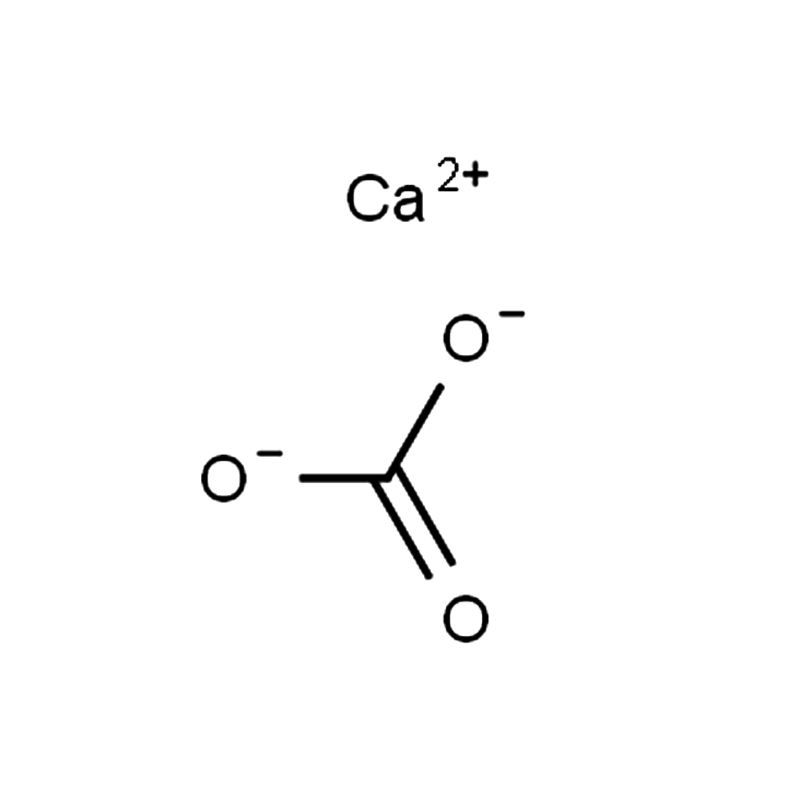
Calcium carbonate CAS#471-34-1
Versatile Natural Mineral Resource – Derived from limestone, marble, chalk, and coral, calcium carbonate is one of the most widely used raw materials in the chemical and building industries.
Essential Building Material Ingredient – Widely applied in the production of cement, mortar, plaster, refractories, and glass, supporting major construction applications.
High-Purity Precipitated Grades Available – Offered as both powdered and precipitated calcium carbonate, with precipitated types providing finer particles, higher purity, and uniform particle size.
Broad Industrial Applications – Used extensively in paper, textiles, paints, plastics, adhesives, sealants, and cosmetics for performance enhancement and quality improvement.
Calcium carbonate CAS#471-34-1
Calcium carbonate is a naturally occurring mineral found in limestone in forms such as marble, chalk, and coral. It is one of the most widely used raw materials in the chemical industry and has extensive applications in construction, including the production of cement, mortar, plaster, refractories, and glass.
It is also an important material for manufacturing quicklime, hydrated lime, and various calcium-based compounds. Calcium carbonate is supplied in both powdered and precipitated forms. Precipitated calcium carbonate features finer particle size, higher purity, and more uniform distribution, making it suitable for a wide range of commercial applications.
Different grades are widely used in industries such as paper, textiles, paints, plastics, adhesives, sealants, and cosmetics.
Calcium carbonate Chemical Properties
| Melting point | 825 °C |
| Boiling point | 800 °C |
| bulk density | 300-1400kg/m3 |
| density | 2.93 g/mL at 25 °C (lit.) |
| refractive index | 1.6583 |
| storage temp. | Store at +5°C to +30°C. |
| solubility | 5 M HCl: 0.1 M at 20 °C, clear, colorless |
| form | random crystals |
| color | White-beige to slightly beige-gray |
| Specific Gravity | 2.93 |
| Odor | Odorless |
| PH | 9.91(1 mM solution);9.91(10 mM solution);9.91(100 mM solution); |
| PH Range | 8 |
| Flame Color | Red-orange |
| Water Solubility | Insoluble |
| λmax | λ: 260 nm Amax: ≤0.09 |
| λ: 280 nm Amax: ≤0.06 | |
| Merck | 141,657 |
| Solubility Product Constant (Ksp) | pKsp: 8.54 |
| BRN | 8008338 |
| Dielectric constant | 6.1(Ambient) |
| Exposure limits | NIOSH: TWA 10 mg/m3; TWA 5 mg/m3 |
| Stability: | Stable. Incompatible with acids, fluorine, ammonium salts, alum. |
| Cosmetics Ingredients Functions | BULKING |
| BUFFERING | |
| ABRASIVE | |
| ORAL CARE | |
| COLORANT | |
| OPACIFYING | |
| InChIKey | VTYYLEPIZMXCLO-UHFFFAOYSA-L |
| Modulus of Elasticity | 10.0 - 80.0 GPa |
| Hardness, Knoop | 75 - 120 |
| Hardness, Mohs | 2.0 - 5.0 |
| Hardness, Shore H | 10 - 60 |
| Hardness, Vickers | 105 - 136 |
| Drilling Hardness | 50 |
| CAS DataBase Reference | 471-34-1(CAS DataBase Reference) |
| EPA Substance Registry System | Calcium carbonate (471-34-1) |
Safety Information
| Hazard Codes | Xi |
| Risk Statements | 37/38-41-36/38-36 |
| Safety Statements | 26-36/37/39-37/39-37 |
| OEB | B |
| OEL | TWA: 10 mg/m3 (total) |
| WGK Germany | - |
| RTECS | FF9335000 |
| TSCA | Yes |
| HS Code | 28365000 |
| Hazardous Substances Data | 471-34-1(Hazardous Substances Data) |
| Toxicity | LD50 orally in Rabbit: 6450 mg/kg LD50 dermal Rat > 2000 mg/kg |
Product Applications of Calcium Carbonate CAS#471-34-1
Calcium carbonate is the calcium salt of carbonic acid and is widely used as an anti-caking agent and dough strengthener. It is available in a range of particle sizes, from coarse granules to fine powder. It is practically insoluble in water and alcohol, although its solubility increases in the presence of ammonium salts or carbon dioxide and decreases in the presence of alkali hydroxides. Its typical pH ranges from 9 to 9.5.
Calcium carbonate is the primary raw material for producing lime (calcium oxide) by heating limestone in a kiln. In food and pharmaceutical applications, it is used as a filler in baking powder, a calcium fortifier, a mild buffering agent in dough, a source of calcium ions in dry dessert mixes, and a neutralizing agent in antacid formulations. It is commonly known as limestone.
Factory and Equipment Display