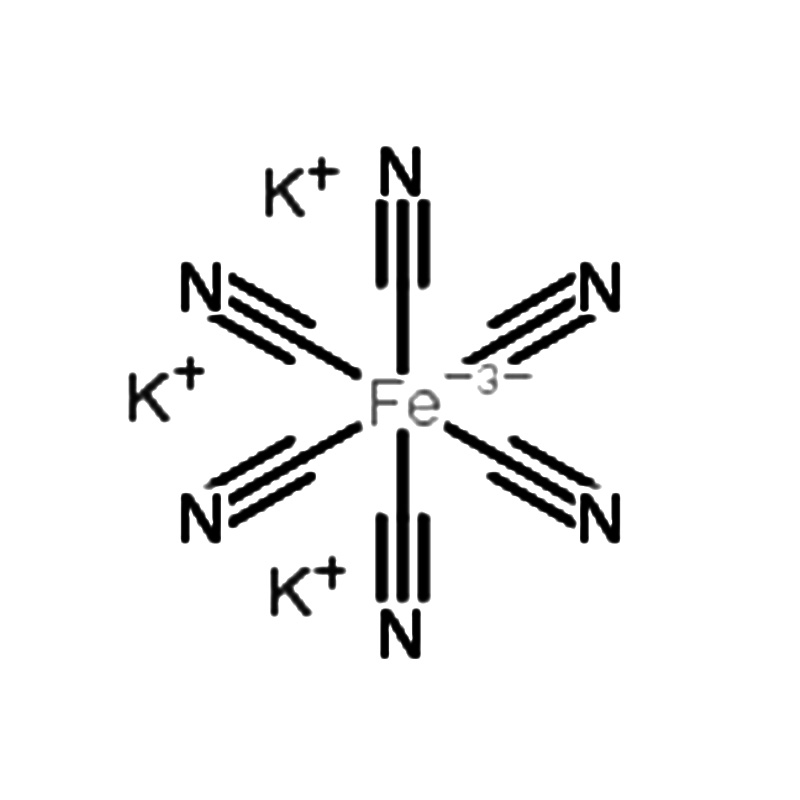
Potassium ferricyanide CAS#13746-66-2
Strong Oxidizing Performance: Potassium ferricyanide acts as an effective oxidizing agent, widely used in photographic processing and chemical reactions.
High Water Solubility: It forms bright red crystals that dissolve easily in water, allowing convenient use in various industrial and laboratory applications.
Versatile Industrial Applications: It is used in pigment production (such as Prussian Blue), metal treatment, electroplating, textile dyeing, and laboratory analysis.
Reliable Redox Control in Experiments: Potassium ferricyanide is commonly used in physiological and chemical experiments to increase and control the redox potential of solutions.
Potassium Ferricyanide CAS# 13746-66-2
Potassium ferricyanide, also known as red prussiate of potash or Prussian red, has the IUPAC name potassium hexacyanoferrate(III). It appears as bright red crystals that are readily soluble in water. The compound is produced by passing chlorine through a solution of potassium ferrocyanide. Although it is generally not considered toxic, it can become hazardous when heated or when mixed with certain other chemicals.
Potassium ferricyanide is commonly used in the production of Prussian Blue, a deep blue pigment widely applied in blueprinting processes. It is also used as a reagent for the detection of ferrous ions. In photographic processing, potassium ferricyanide serves as an oxidizing agent to remove silver from negatives and positives through a process known as dot etching. In color photography, it can reduce the size of color dots without decreasing their number, functioning as a form of manual color correction.
Additionally, potassium ferricyanide is frequently used in physiological and chemical experiments to increase the redox potential of solutions (E° ≈ 436 mV at pH 7). It is also applied in metal treatment processes, including the hardening of iron and steel, as well as in electroplating, wool dyeing, and laboratory reagent applications.
Potassium ferricyanide Chemical Properties
| Melting point | °Cd ec.) |
| Bulk density | 900-1000kg/m3 |
| Density | 1.85 |
| Vapor pressure | 0Pa at 20℃ |
| Storage temp | Store at +5°C to +30°C. |
| Solubility | H2O: 1 M at 20 °C, complete, orange-brown |
| Form | fine crystals |
| Color | Orange to red |
| Specific Gravity | 1.88 |
| Odor | Odorless |
| PH | 6-9 (25℃, 1M in H2O) |
| PH Range | 6 - 9 at 329 g/l at 25 °C |
| Water Solubility | 464 g/L (20 ºC) |
| Sensitive | Light Sensitive |
| Merck | 147630 |
| Exposure limits | ACGIH: TWA 1 mg/m3 |
| NIOSH: IDLH 25 mg/m3; TWA 1 mg/m3 | |
| Stability | Stable. Incompatible with ammonia, strong acids, strong oxidizing agents. Reaction with acids yields toxic gas. May discolour on exposure to light. |
| InChI | 1S/6CN.Fe.3K/c6*1-2;;;;/q;;;;;;-3;3*+1 |
| InChIKey | MIMJFNVDBPUTPB-UHFFFAOYSA-N |
| SMILES | [K+].[K+].[K+].N#C[Fe-3](C#N)(C#N)(C#N)(C#N)C#N |
| CAS DataBase Reference | 13746-66-2(CAS DataBase Reference) |
| EPA Substance Registry System | Ferrate(3-), hexakis(cyano-.kappa.C)-, potassium (1:3), (OC-6-11)- (13746-66-2) |
| Absorption | no transmittance (UV) in H2O at 1M |
Safety Information
| Hazard Codes | Xn |
| Risk Statements | 32-20/21/22 |
| Safety Statements | 50A-36 |
| RIDADR | UN 3077 9 / PGIII |
| WGK Germany | 2 |
| RTECS | LJ8225000 |
| F | 8 |
| TSCA | TSCA listed |
| HS Code | 28372000 |
| Storage Class | 11 - Combustible Solids |
| Hazard Classifications | Aquatic Chronic 2 |
| Eye Irrit. 2 |
Product Application of Potassium Ferricyanide CAS# 13746-66-2
Potassium ferricyanide is widely used in amperometric biosensors as an electron transfer mediator. In these systems, it replaces the enzyme’s natural electron transfer agent when working with enzymes such as glucose oxidase, helping to facilitate efficient electron transfer during detection.
In addition, potassium ferricyanide is an important component of Murakami’s etchant, which is commonly used for the etching and analysis of cemented carbides in metallurgical applications.